Part 7: Analytical Techniques for Stereochemistry
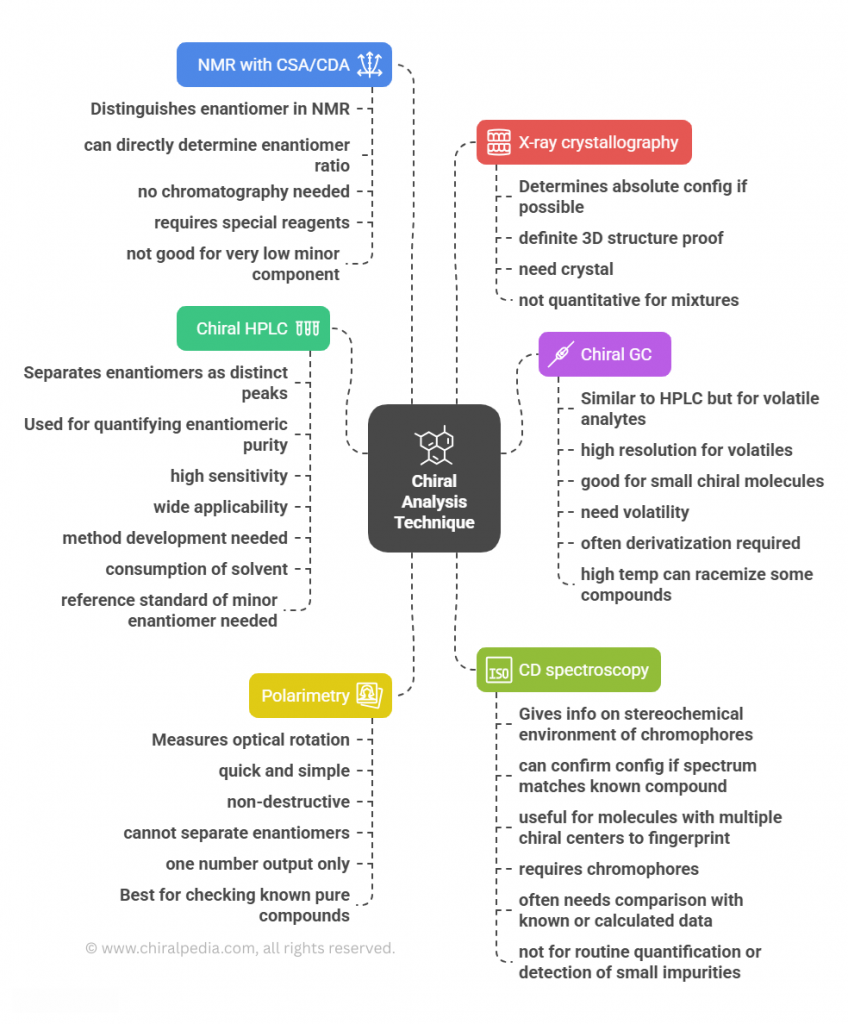
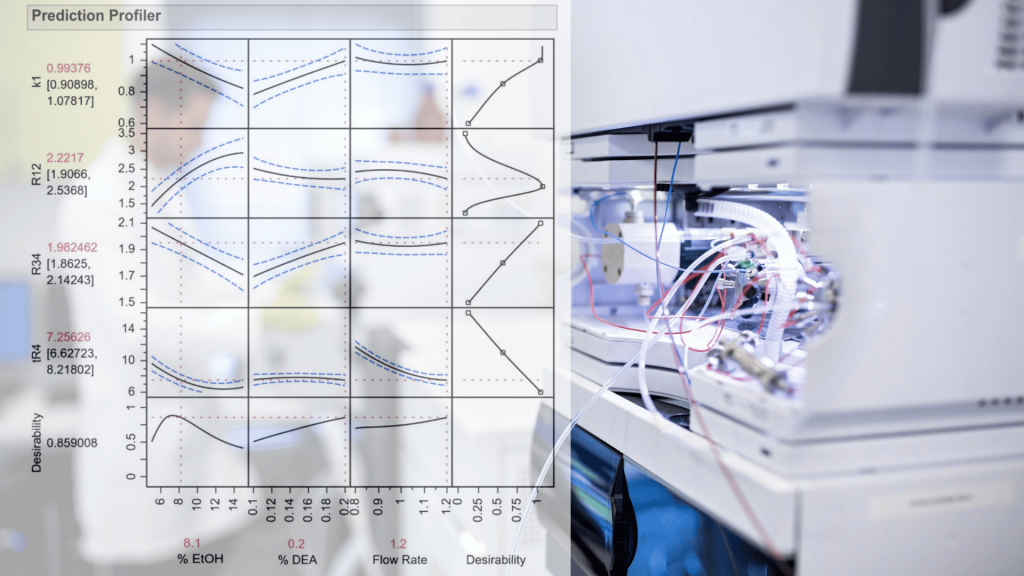
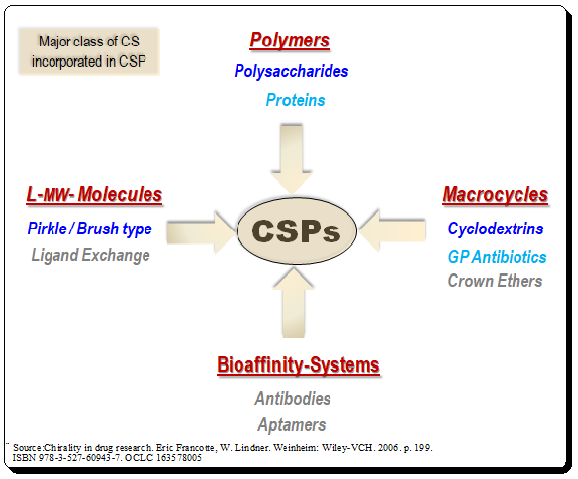
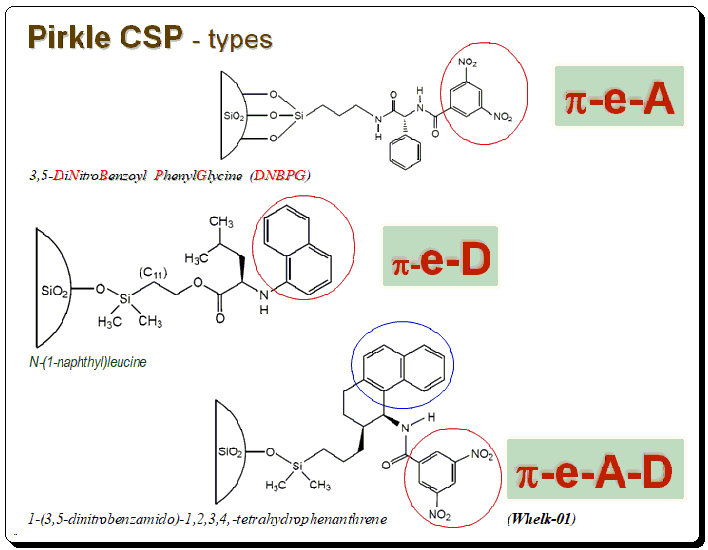
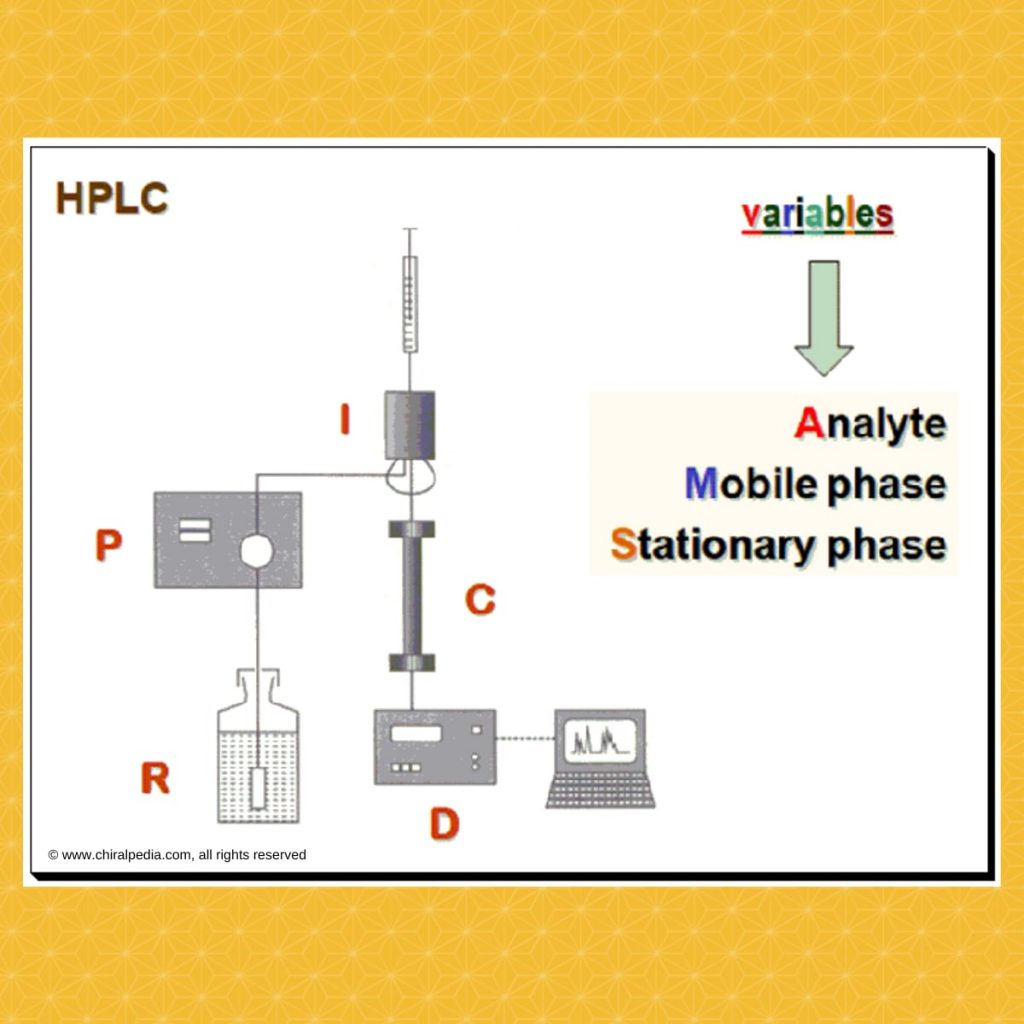
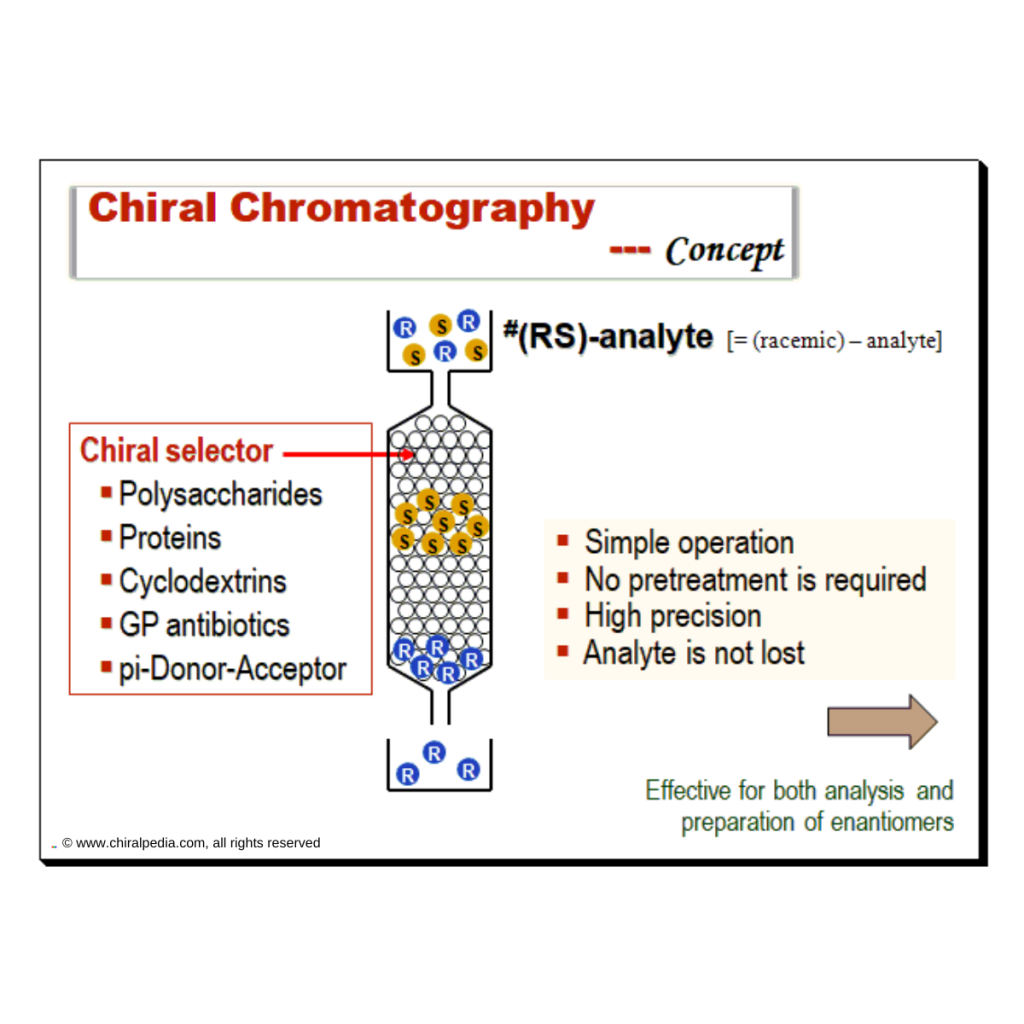
“Measuring handedness – tools and techniques that bring chirality into focus” Introduction Ensuring the correct stereochemistry and measuring stereochemical purity is a crucial aspect of pharmaceutical quality control and research. This part covers the major analytical techniques used to distinguish and quantify enantiomers and diastereomers in drug substances and products. Key techniques include: We’ll discuss how these techniques are applied in practice: e.g., during process development, a chiral HPLC method is developed to monitor enantiomeric …
Part 7: Analytical Techniques for Stereochemistry Read More »