
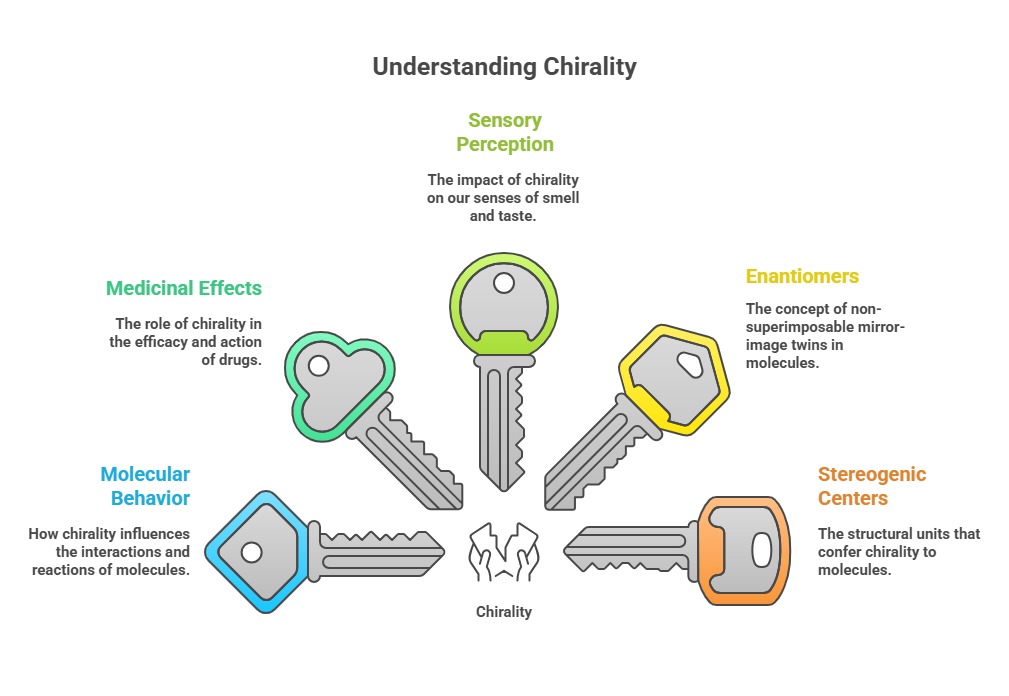
Chirality: it's everywhere - shaping how molecules behave, how medicines work, and even how scents and flavors hit our senses. Chirality is a property of an object which is non-superimposable on its mirror image. The word chiral stems from the Greek word “cheir”, which means 'handedness'. When an object cannot be superimposed on its mirror image, such a pair is referred to as chiral. It is just like the left and right hands. The two non-superimposable mirror-image twins of a chiral molecule is called enantiomers. The most commonly encountered stereogenic unit that confers chirality to drug molecules is stereogenic center. Stereogenic center can be due to the presence of tetrahedral tetra coordinate atoms (C,N,P) and pyramidal tricoordinate atoms (N,S). The word chiral describes the three-dimensional architecture of the molecule and does not reveal the stereochemical composition.
"From atoms to human beings, nature is asymmetric with respect to chirality, or left- and right-handedness. Clues are beginning to emerge that connect chirality on different levels" ...
"All life on Earth is made of molecules that twist in the same direction. New research reveals that this may not always have been so" ...
"The mirror-image asymmetry of life is one of the biggest mysteries in biology" ...
Roger A. Hegstrom and Dilip K. Kondepudi
Scientific American, January, 108-115, 1990.
https://quantummechanics.ucsd.edu/ph87/ScientificAmerican/Sciam/Hegstrom_The_Handedness_of_the_universe.pdf
![]() https://www.researchgate.net/publication/234614382_The_Handedness_of_the_Universe
https://www.researchgate.net/publication/234614382_The_Handedness_of_the_Universe
![]()
Rachel Brazil
Chemistry World, 26, October, 2015
https://www.chemistryworld.com/features/the-origin-of-homochirality/9073.article
![]()
Maxim Papusha and Kai Leonhard
Journal of Chemical Information and Modeling, 2026.
Yichuan Peng, Gufeng Yu, Runhan Shi, Letian Chen, Xi Wang, Wenjie Du, Xiaohong Huo, Yang Yang.
Artificial Intelligence Chemistry, 3, 2, December, 2025, 100091.