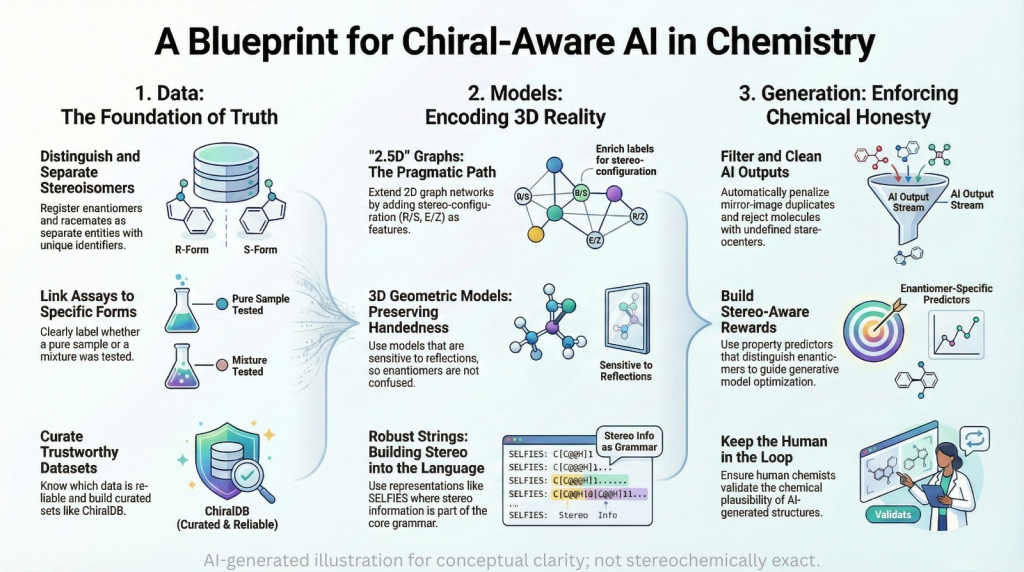
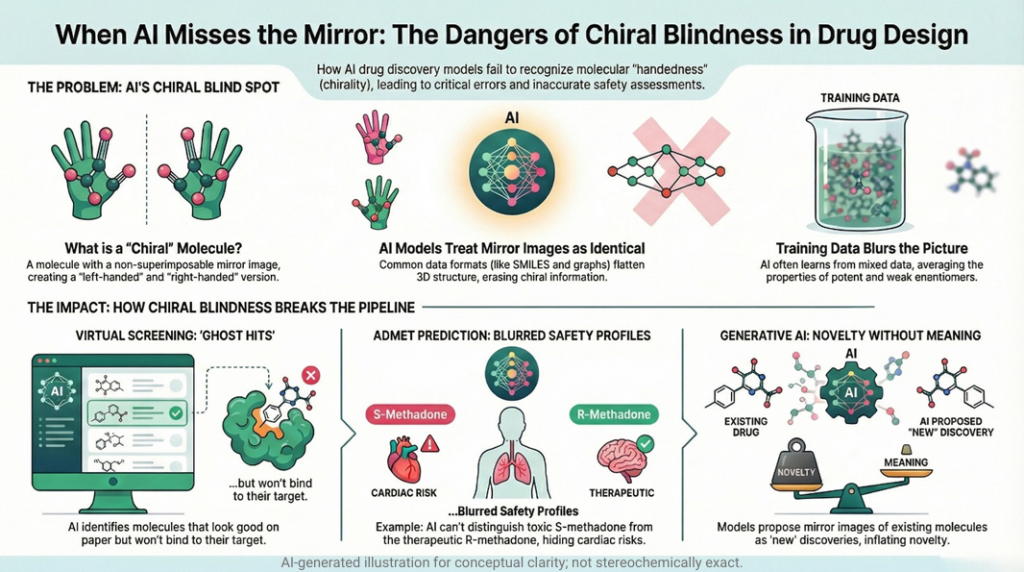
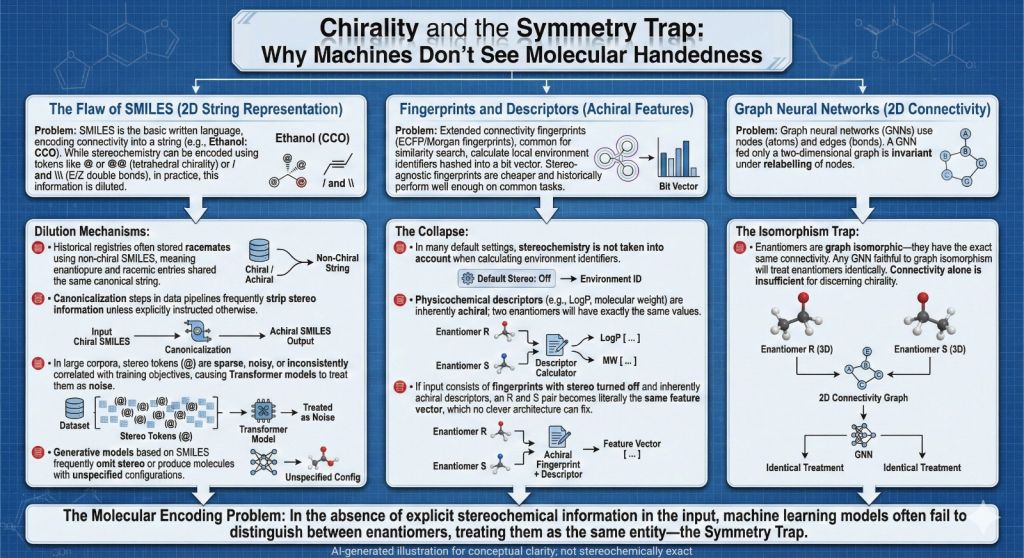
Germanium 🧬: Beyond Carbon — A Chiralpedia Guide to a New Dimension of Chirality
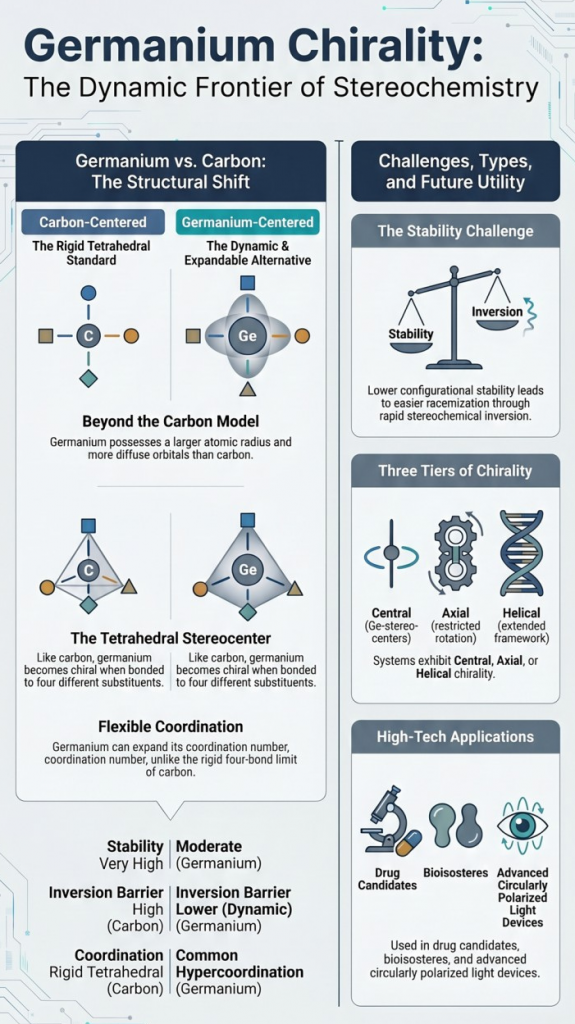
Demonstrates that chirality extends beyond carbon into heavier elements Introduction: Beyond Carbon-Centered Chirality Chirality is one of the most fundamental concepts in chemistry, typically introduced through tetrahedral carbon atoms bearing four different substituents. However, chirality is not limited to carbon—it extends across the periodic table. Germanium, a Group 14 element positioned below silicon, presents a fascinating case. While it shares structural similarities with carbon, its larger atomic size, flexible coordination, and unique electronic structure give …
Germanium 🧬: Beyond Carbon — A Chiralpedia Guide to a New Dimension of Chirality Read More »