Molecular Handedness: How Chirality Shapes Molecules
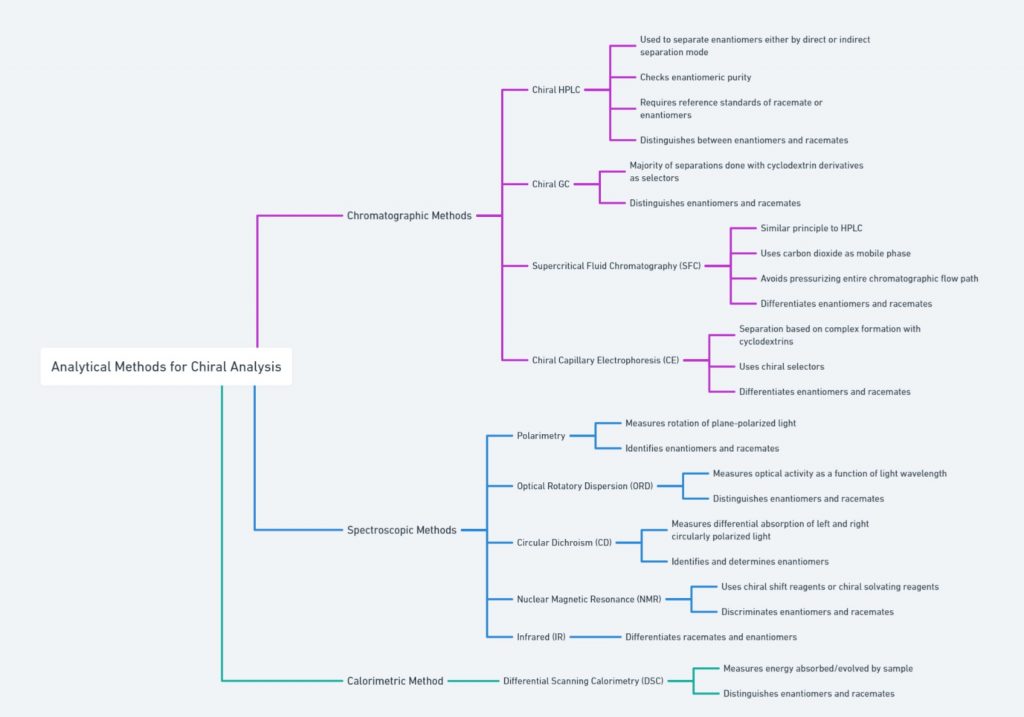
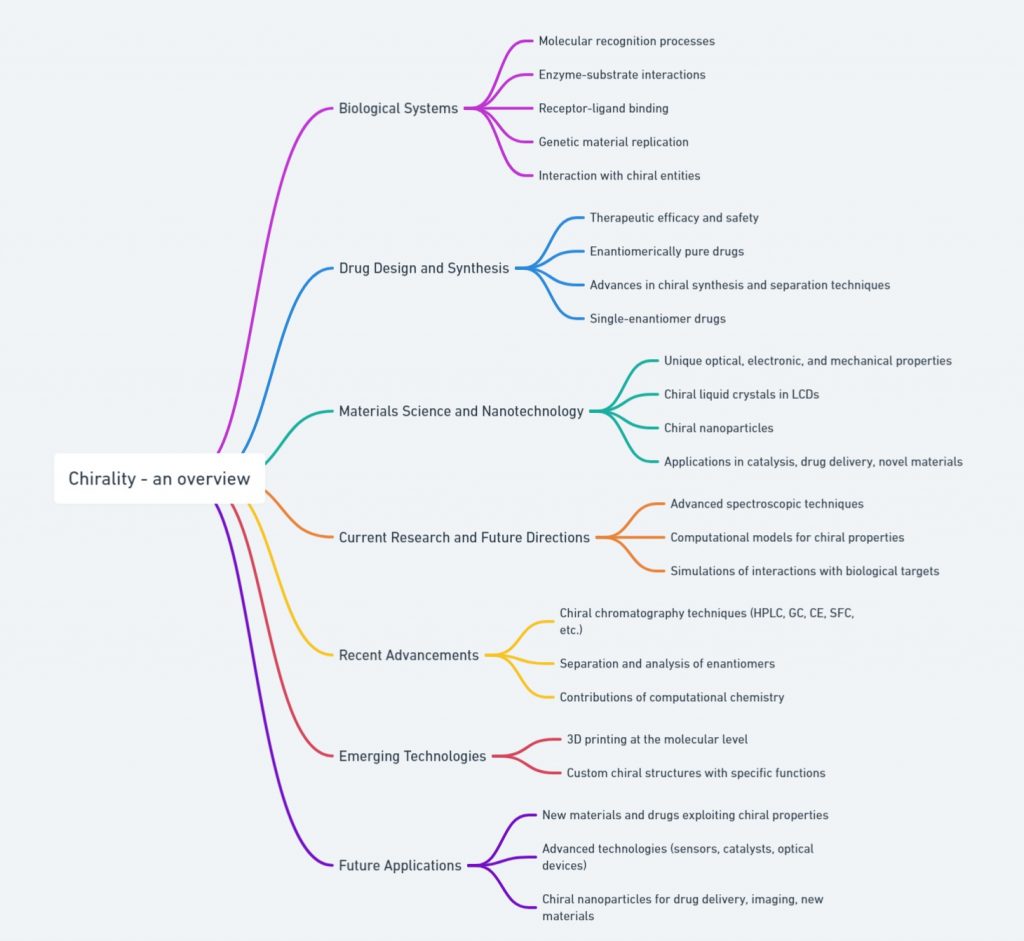
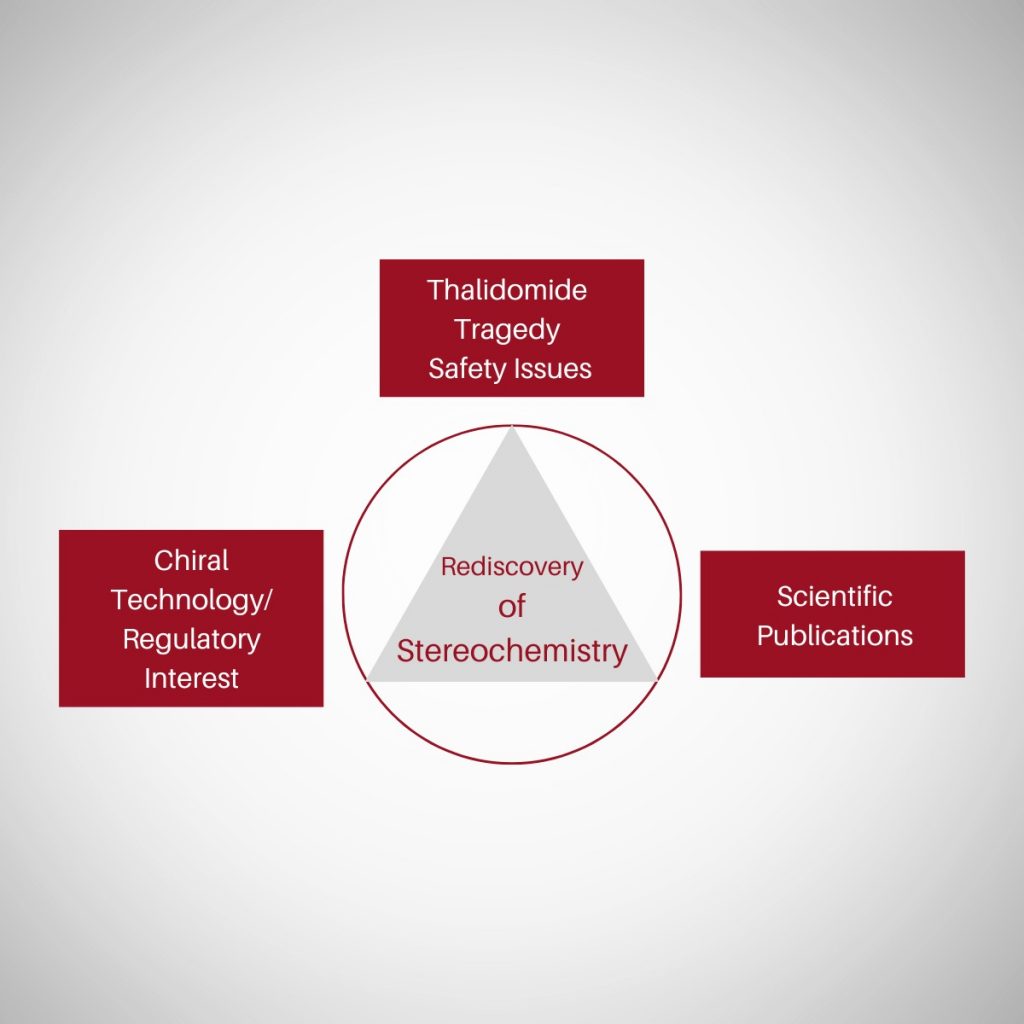
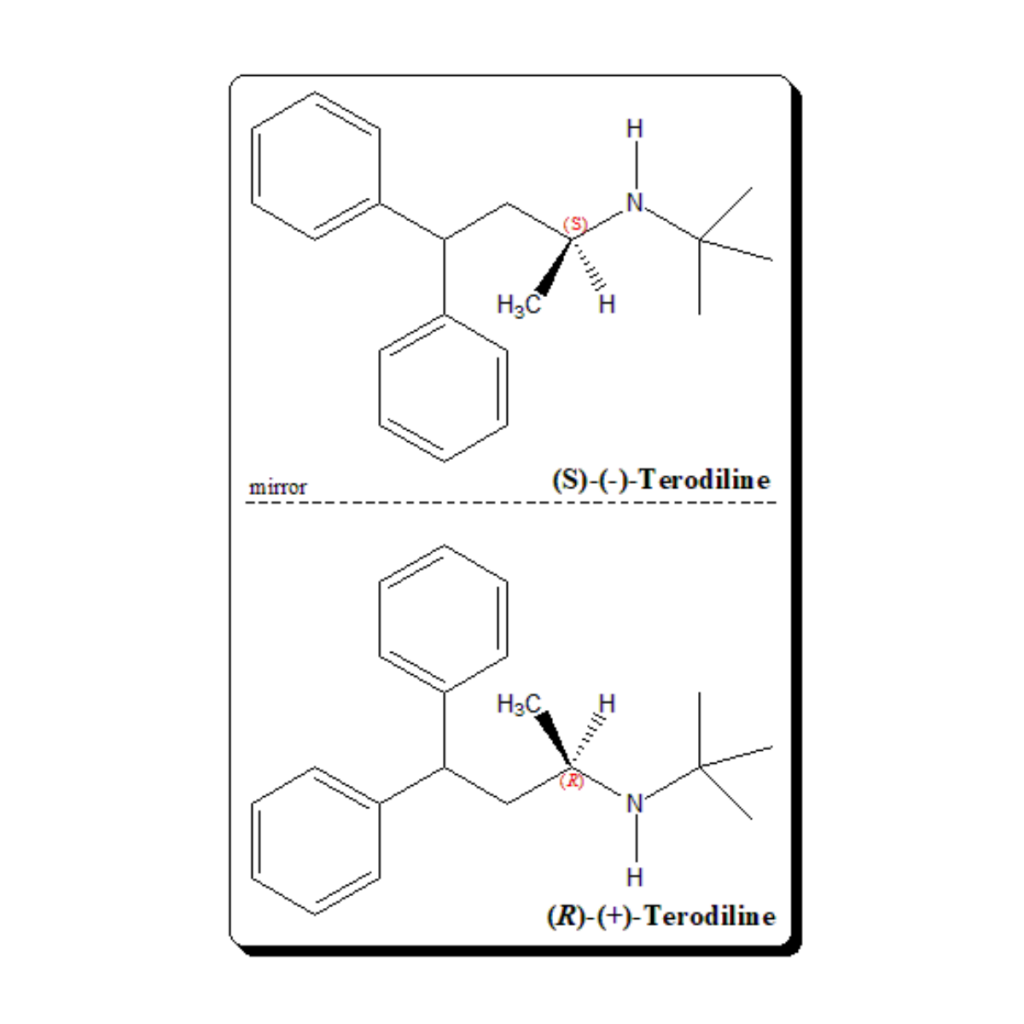
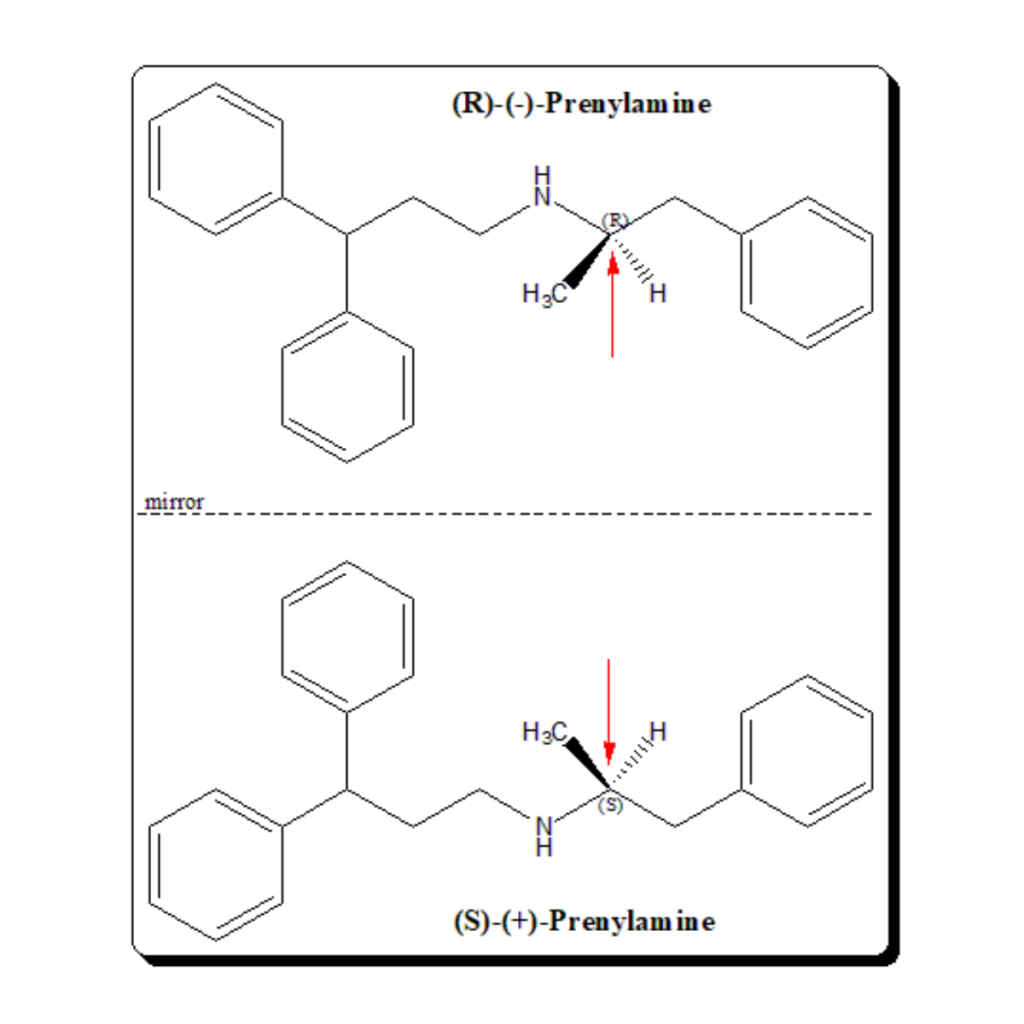
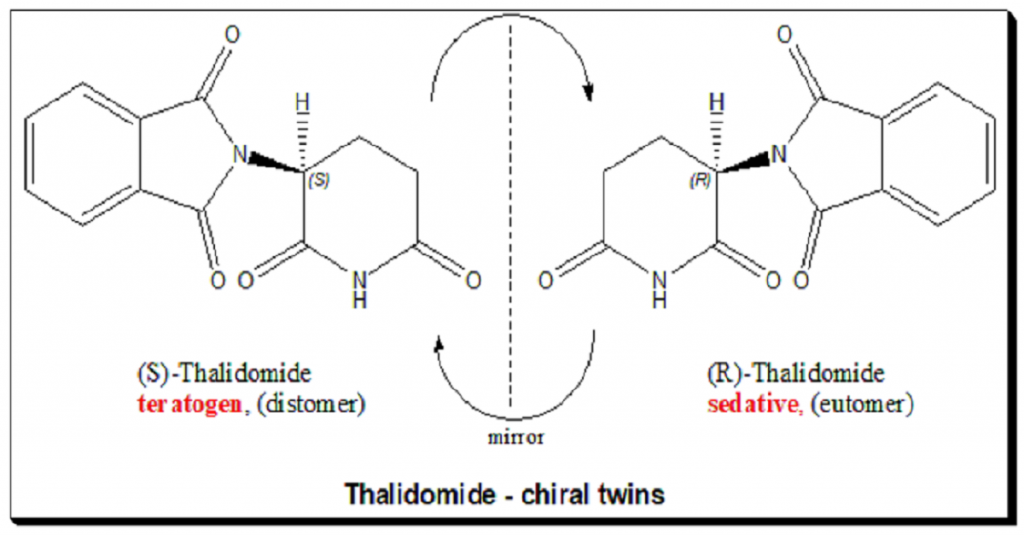
Introduction Molecules can come in two shapes that mirror each other, in the same way as our left and right hands. This attribute, called chirality, can be found in biological molecules like sugars and proteins This property, often referred to as molecular handedness, plays a crucial role in chemistry, biology, and pharmacology. In this article, we …
Molecular Handedness: How Chirality Shapes Molecules Read More »