“From left- and right-handedness to life’s molecular signatures—chirality explained”
Introduction
Building on the overview of chirality, this section delves into core concepts: symmetry elements in molecules, the definitions of enantiomers and diastereomers, and the phenomenon of optical activity. Understanding these fundamentals is essential for grasping how stereochemistry manifests and is measured. We will also explore how chirality is quantified via optical rotation and how instruments like polarimeters help distinguish enantiomers. By the end of this part, readers should be comfortable with terms like enantiomer, diastereomer, racemate, specific rotation, and polarimetry, setting the stage for stereochemical nomenclature in Part 3.
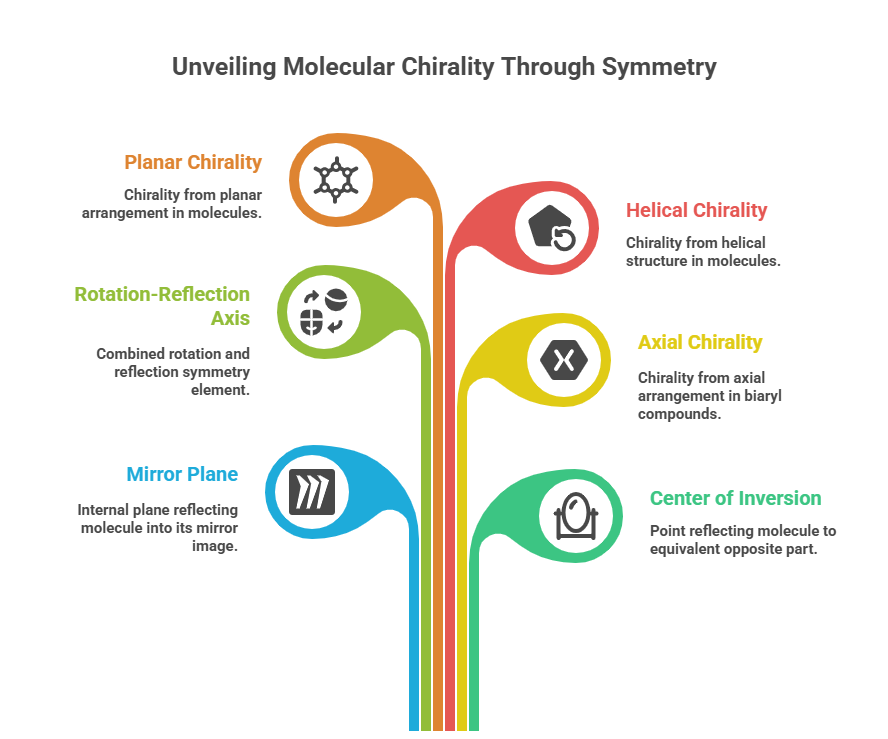
Elements of Symmetry and Chirality
A molecule’s chirality is intimately related to its symmetry (or lack thereof). The presence of certain symmetry elements will render a molecule achiral, even if it contains stereocenters. Key symmetry elements to consider are:
– Mirror Plane (σ): An internal plane that reflects half of the molecule into the other half. If a molecule has a mirror plane, it is superimposable on its mirror image (hence achiral). For example, meso-tartaric acid has two stereocenters, but also an internal mirror plane, making it achiral despite chiral centers.
– Center of Inversion (i): A point through which all parts of the molecule reflect to an equivalent opposite part. Molecules with a center of inversion are also achiral.
– Rotation-Reflection Axis (S<sub>n</sub>): A combined symmetry of rotation followed by reflection. Particularly, an S<sub>2</sub> is equivalent to an inversion center, and an S<sub>1</sub> is a mirror plane. Chirality is often characterized as the absence of any S<sub>n</sub> axis; a chiral molecule cannot have an S<sub>n</sub> symmetry element for any n.
In practice, a quick test for chirality is to look for an internal mirror plane: if one exists, the molecule is achiral (or meso). If not, and the molecule is not identical to its mirror image, it’s chiral. For instance, 2-butanol is chiral (no symmetry plane, one stereocenter), whereas 2,3-butanediol has stereocenters but one stereochemical configuration (meso form) possesses a mirror plane, making it achiral. Ethambutol, an antitubercular agent, exists in multiple stereoisomeric forms – among them, the meso-isomer, distinguished by its internal plane of symmetry. This symmetry renders it achiral, despite having stereocenters. < To explore the concept further checkout chiralpedia blog on meso-compounds and plane of symmetry @ <https://chiralpedia.com/blog/the-meso-compounds-finding-plane-of-symmetry/>.
Thus, chirality can arise not only from single stereocenters but also from axial chirality (as in biaryl compounds like certain drugs or natural products), planar chirality, and helical chirality – none of which have symmetry elements that make them superimposable on their mirror forms. These advanced types will be touched upon later in the series; the unifying principle is the absence of improper symmetry that connects a molecule to its mirror image. For a detailed discussion on axial chirality (atropisomerism) refer to the chiralpedia blog @ <https://chiralpedia.com/blog/atropisomers-things-are-tight-single-bond-wont-rotate/>.
Enantiomers and Diastereomers
Enantiomers are a pair of stereoisomers that are non-superimposable mirror images of each other. They have identical connectivity and (in the absence of chiral environments) identical physical properties (melting point, boiling point, NMR spectra, etc.), except for how they interact with plane-polarized light and other chiral substances. A classic example is the R- and S-enantiomers of limonene.

Another example: D-glucose and L-glucose are enantiomers; our metabolic enzymes (which are chiral) can metabolize D-glucose readily, whereas L-glucose is not recognized (and is essentially calorie-free). Importantly, enantiomers rotate plane-polarized light in equal magnitude but opposite directions (more on this below). In pharmacology, enantiomers often have different activities (e.g., one may fit a receptor better). [Read more @ <https://chiralpedia.com/blog/chiral-pharmacology-the-mirror-image-of-drug-development/>. Enantiomers are sometimes designated as “left-” or “right-handed” forms of a molecule.
Diastereomers
Diastereomers are stereoisomers that are not mirror images of each other. This situation arises when molecules have multiple stereocenters. Two examples are presented to illustrate this concept. Tartaric acid and Ephedrine.
Illustration 1: Tartaric acid
Tartaric acid: it has two stereocenters. The molecule can exist as RR, SS (which are enantiomers of each other), and RS (which is a meso-form identical to SR after rotation, and achiral). The RR vs. RS forms are diastereomers – they are both stereoisomers of tartaric acid but are not mirror images. Diastereomers have different physical and chemical properties (unlike enantiomers).
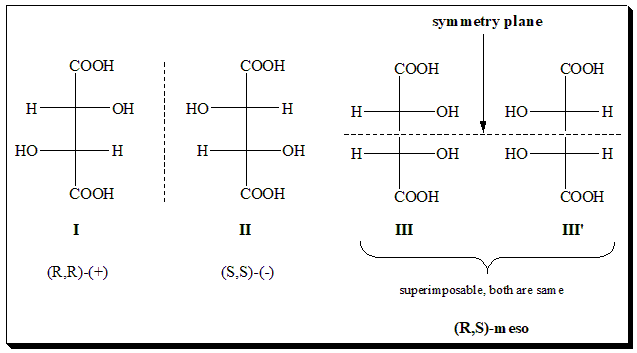
I and III are diastereomers. For a more details consult the blog @ <https://chiralpedia.com/blog/the-meso-compounds-finding-plane-of-symmetry/>
Illustration 2: Ephedrine and pseudoephedrine.
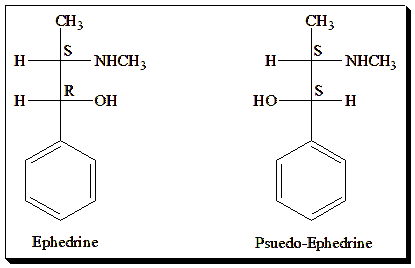
In pharmaceuticals, diastereomers can often be separated by conventional means (because of differing solubilities, etc.), and if both are biologically active, they might be developed as separate drugs or one chosen over the other. Geometric isomers (like cis/trans or E/Z isomers of double bonds) are also a type of diastereomer <For an in-depth look check out the full blog article @ https://chiralpedia.com/blog/cis-trans-and-e-z-notation-choose-your-side/>.
The FDA guidance explicitly notes that geometric isomers and diastereomers should be treated as distinct drugs unless they interconvert in vivo, because they often have distinct pharmacology. A poignant example: cis-platin (cis-diamminedichloroplatinum(II)) is a potent anticancer drug, whereas the trans isomer (trans-platin) is ineffective as an anticancer agent – a clear case where geometric diastereomers have different biological effects.
Racemic Mixtures
Racemic Mixtures: A 50:50 mixture of enantiomers is called a racemate or racemic mixture (sometimes denoted with a (±) prefix). Racemates are overall optically inactive (the two enantiomers’ rotations cancel out). However, racemates can have different properties from either pure enantiomer. For instance, a racemic compound might crystallize in a different form than enantiopure samples (some racemates form a distinct crystal lattice containing both enantiomers). In drug development, a racemic mixture might be easier or harder to crystallize or purify than a single enantiomer, so sometimes a racemate is used in a formulation by design (we will discuss the pros and cons in Part 6). An interesting note: some racemic mixtures can undergo spontaneous resolution upon crystallization, where enantiomers crystallize separately – a phenomenon Pasteur exploited. But in general, separating a racemate into enantiomers (resolution) is non-trivial.
Optical Activity and Specific Rotation
One of the earliest methods to characterize chirality is through a property called optical activity. A substance is optically active if it can rotate the plane of plane-polarized light. Enantiomers rotate light to an equal degree but in opposite directions: one is dextrorotatory (rotates light clockwise, denoted “+” or “d”), and the other is levorotatory (rotates light counterclockwise, denoted “–” or “l”). Notably, the direction (+/–) of optical rotation is entirely empirical and is not directly related to the R/S configuration (which is determined by structure, as we’ll see in Part 3). For example, (R)-limonene is (+) (smells like orange), whereas (S)-limonene is (–) but for other molecules, (R)- could be (–) and S-(+), there’s no universal correlation.
- Polarimetry: The instrument used to measure optical rotation is a polarimeter. It consists of a light source emitting monochromatic light (typically sodium D line at 589 nm), a polarizer to create plane-polarized light, a sample tube, and an analyzer (a second polarizing filter that can be rotated). When an optically active sample is in the tube, the plane of polarization is rotated by some angle α. The analyst rotates the analyzer until light passes through with maximum intensity; the angle needed is the optical rotation. Polarimetry is often done at a specified temperature (usually 20 °C) and wavelength (the sodium D line, labeled as the subscript D). The specific rotation [α] is defined as the standardized rotation angle for a sample with a path length l (usually 1 decimeter) and concentration c (in g/mL for solutions, or using density for neat liquids). Mathematically,
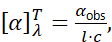
where T is temperature (°C) and λ is the wavelength used (often the D line). For example, if a 1 dm tube containing a solution of 1 g/mL of a chiral compound produces an observed rotation of +12°, the specific rotation [α]<sup>20</sup><sub>D</sub> = +12° (by definition under those conditions). If the tube length or concentration differ, one computes [α] by scaling the observed α. Specific rotation is an intrinsic property of a chiral substance (at a given wavelength and temperature) – an identity parameter that can be compared to literature values to confirm purity or absolute configuration. E.g., pure (S)-ibuprofen has a specific rotation of about +54° (in methanol, 20 °C, D line), whereas the racemic ibuprofen would have [α] ≈ 0° because the contributions cancel (external compensation).
- Optical Activity Significance: Optical rotation was historically crucial: it was evidence of molecular asymmetry before X-ray crystallography existed. Pasteur’s separated tartaric acid enantiomers exhibited equal and opposite rotations, proving enantiomers’ existence. In modern pharma labs, polarimetry is still used for quick checks of enantiomeric purity or identity. However, it provides no detail on which enantiomer (R or S) you have – that requires knowing the absolute configuration by other methods (see Part 3 and Part 8).
- Polarimetry in Practice: Consider that many early drugs were characterized by optical rotation. L-epinephrine (adrenaline) was identified as levorotatory, and its specific rotation is part of pharmacopoeia standards. If a batch deviated, that might indicate racemization or impurity. Even today, pharmacopoeias list [α] for chiral drugs as an identification criterion.
Polarimetry and Chirality Quantification
A key term is enantiomeric excess (ee), which quantifies optical purity. It’s defined as . A racemate has 0% ee, an enantiomerically pure sample has 100% ee. If you measure an optical rotation that is 50% of the literature value for the pure enantiomer, the sample is presumably a 75:25 enantiomeric mixture (50% ee). Modern chiral analysis (see Part 8) often uses chiral chromatography or NMR, but optical rotation provides a quick estimate of enantiomeric composition if [α] of the pure enantiomers is known.
Enantiomers in a Chiral Environment
While enantiomers share properties in achiral environments, they behave differently in chiral settings. Two striking examples in a biological context:
– Carvone enantiomers: R-(–)-carvone smells like spearmint, S-(+)-carvone smells like caraway. Our nose’s receptors are chiral protein pockets that distinguish them.
– Drug receptors: Thalidomide exists as an enantiomeric pair. The (R)-enantiomer has the desired sedative effect while the (S)-enantiomer harbors embryo-toxic and teratogenic effect. The enantiomers of the beta-blocker propranolol, (S)-propranolol is found to be 130 times as active as its (R)-enantiomer. Another case is warfarin, an anticoagulant. Warfarin’s enantiomers are both anticoagulant, but S-warfarin is ~4 times more potent and is metabolized by a different enzyme than R-warfarin. This affects dosing and drug interactions – a quintessential demonstration that even when both enantiomers are “active,” their pharmacokinetics and dynamics can diverge.
Diastereomers in Pharmacy
Diastereomers, because they have different shapes (not mirror images), can bind to targets in unrelated ways or be processed differently. For example, the antidepressant venlafaxine has a chiral center and is used as a racemate. Its O-desmethyl metabolite (ODV) is also chiral. One diastereomeric pair (venlafaxine + ODV) might be more active or longer-lived than the other. While this is complex, it reminds us that when multiple stereocenters are in play, the combinatorial possibilities (2n) stereoisomers for n centers) include many diastereomers, each potentially a unique chemical entity. Regulatory guidance is to treat each as separate unless interconversion is proven.
Summary (Part 2)
– Chirality and Symmetry: Chiral molecules have no symmetry that makes them superimposable on their mirror image (no mirror plane, inversion center, etc.). Achiral molecules either lack stereocenters or have symmetry (like meso compounds) that renders them superimposable on the mirror image.
– Enantiomers: Stereoisomers that are mirror images (e.g. D- vs L-glucose). They share most properties except interactions with other chiral entities (including plane-polarized light and chiral receptors/solvents). Enantiomers rotate plane-polarized light in equal and opposite directions.
– Diastereomers: Stereoisomers that are not mirror images (e.g. cis- vs trans-2-butene, or threo- vs erythro-diastereomers).[For more detailed discussion on this nomenclature consult the blog article @ <https://chiralpedia.com/blog/erythro-and-threo-prefixes-the-same-or-opposite-side/>].Consult They have different physical properties and can be separated more easily. Geometric isomers and molecules with multiple stereocenters may produce diastereomers.
– Optical Activity: A hallmark of chirality. Measured by polarimetry, quantified as specific rotation [α]. Enantiomer pairs have opposite rotations. Racemic mixtures are optically inactive overall.
– Polarimetry: Important for characterizing chiral compounds. Historically crucial, and still useful for quick purity checks. However, it doesn’t identify which enantiomer is which (that requires knowing absolute configuration or comparing rotation sign to literature).
– Pharmaceutical examples: Many chiral drugs illustrate these concepts: one enantiomer of a drug can be therapeutically active while the other is less active or causes side effects (e.g., d- vs l-propranolol, thalidomide enantiomers. Diastereomeric drugs (like cis/trans isomers or compounds with >1 stereocenter) likewise may have one form approved and the other not, due to efficacy or safety differences.
Suggested Reading
IUPAC Compendium of Chemical Terminology (Gold Book): Definitions of enantiomer, diastereoisomer, optical rotation, racemate. (Provides clear, authoritative definitions.)
Organic Chemistry (by Clayden, Greeves, Warren, Wothers), Chapter on Stereochemistry. (An accessible discussion of symmetry elements, enantiomers vs diastereomers, and optical activity with many illustrations.)
https://en.wikipedia.org/wiki/Atropisomer
https://chem.libretexts.org/Bookshelves/Organic_Chemistry/Organic_Chemistry_(OpenStax)/05%3A_Stereochemistry_at_Tetrahedral_Centers/5.04%3A_Pasteur’s_Discovery_of_Enantiomers (Narrates Pasteur’s experiment in detail, connecting optical activity with molecular chirality.)
P. Y. Wang, et al. (1980). Science, 209, 1420-1421. (Brief report on D- vs L-glucose metabolism, illustrating enantiomer recognition in biology.)
Health Canada Guidance “Stereochemical Issues in Chiral Drug Development” (2000). Sections on terminology and analysis. (A regulatory perspective on defining and handling stereochemical forms, reinforcing many concepts in this part.)
Development of New Stereoisomeric Drugs, 1992. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/development-new-stereoisomeric-drugs
https://www.pharmabiz.com/NewsDetails.aspx?aid=162712&sid=9#
https://en.wikipedia.org/wiki/Chiral_drugs