Why stereochemistry is not just about seeing molecules—but learning how to describe them
In modern chemistry, we have powerful tools like X-ray crystallography that can reveal the exact three-dimensional structure of a molecule with remarkable precision. So it raises a compelling question:
If we already know what a molecule truly looks like in 3D… why do we still assign R/S configuration on a flat 2D drawing?
Is it redundancy—or something deeper?
🔬 Discovery vs Description
The answer lies in a subtle but powerful distinction: knowing a structure is not the same as communicating it.
X-ray crystallography gives us the absolute spatial arrangement of atoms—the truth of the molecule.
But chemistry does not live in instruments alone. It lives in:
- research papers
- lab notebooks
- textbooks
- digital screens
All of which are basically 2D representations.
✏️ The Language of Chirality
This is where R/S configuration becomes indispensable.
It is not an experimental result, but a universal language—a standardized way to translate a 3D arrangement into an unambiguous descriptor.
Without R/S:
- The same molecule can be drawn in multiple valid ways
- Interpretations may differ
- Communication becomes uncertain
With R/S:
- Every chemist, anywhere in the world, can reconstruct the exact same 3D structure from a 2D drawing
🧠 A Simple Way to See It
Think of it like this:
📍X-ray crystallography is like physically visiting a location and mapping it precisely
📍R/S nomenclature is like writing down its address
Even if you know the place exists, you still need an address to communicate it clearly.
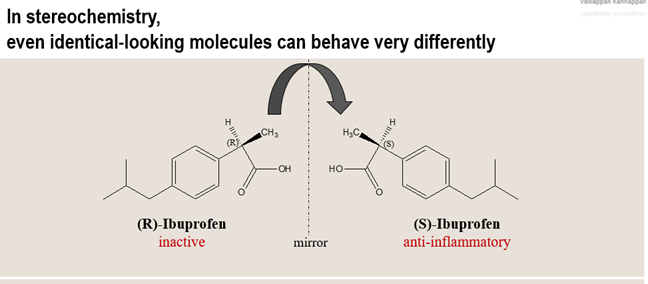
💊 Why This Matters
In real-world chemistry, especially in drug development, this clarity is critical. Take Ibuprofen as an example:
one enantiomer is biologically active, while the other is far less effective.
🎯 The Takeaway
R/S assignment is not about discovering structure. It is about standardizing how we describe it.
X-ray reveals the truth.
R/S ensures that truth can be clearly spoken, shared, and understood.
✨ Through the Chiralpedia Lens
Chirality is often taught as a visual concept—wedge bonds, dashed lines, and 3D imagination. But its real power lies beyond visualization. It lies in the ability to:
- think structurally
- describe precisely
- communicate universally
Because in chemistry, discovery is only the beginning—
understanding grows when we can express what we see without ambiguity.

Further Reading

