Demonstrates that chirality extends beyond carbon into heavier elements
Introduction: Beyond Carbon-Centered Chirality
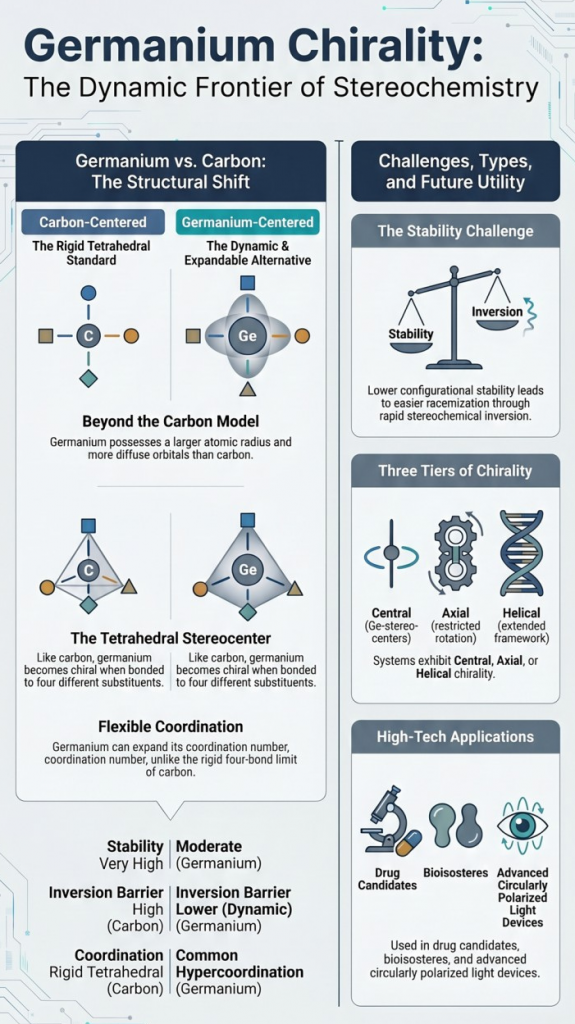
Chirality is one of the most fundamental concepts in chemistry, typically introduced through tetrahedral carbon atoms bearing four different substituents. However, chirality is not limited to carbon—it extends across the periodic table.
Germanium, a Group 14 element positioned below silicon, presents a fascinating case. While it shares structural similarities with carbon, its larger atomic size, flexible coordination, and unique electronic structure give rise to distinct stereochemical behavior.
This tutorial introduces the principles of germanium-centered chirality, explores how it differs from carbon, and highlights why it is becoming increasingly relevant in modern chemistry.
1. Why Germanium Can Be Chiral
Germanium commonly forms tetrahedral compounds (GeR₄), analogous to carbon. When bonded to four different substituents, germanium becomes a stereogenic center, giving rise to two enantiomers.
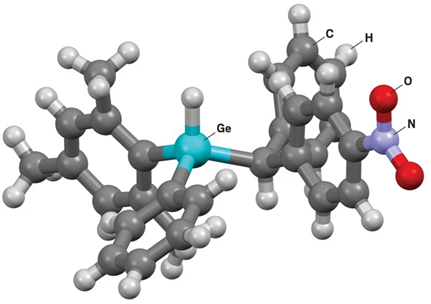
This follows the general definition of chirality as a non-superimposable mirror-image relationship, applicable across organic and inorganic systems.
However, germanium differs from carbon in several ways:
- Larger atomic radius
- More diffuse orbitals
- Increased polarizability
These factors influence both reactivity and stereochemical behavior.
2. Assigning Configuration at Germanium
The assignment of absolute configuration at germanium follows the Cahn–Ingold–Prelog (CIP) rules, just as for carbon. Read more @ <https://chiralpedia.com/blog/naming-enantiomers-the-r-s-system/>
Key steps:
- Rank substituents by atomic number
- Orient the lowest priority group away
- Determine the sequence (clockwise = R, counterclockwise = S)
Because germanium often bonds to heavier atoms, priority assignments can involve higher atomic numbers, making stereochemical ordering slightly less intuitive than in carbon systems.
3. Stability of Germanium Chirality: A Key Challenge
A defining feature of germanium chirality is its lower configurational stability compared to carbon.
- Carbon stereocenters: highly stable
- Germanium stereocenters: more prone to racemization
This is because germanium can:
- Expand its coordination number
- Form hypercoordinate (5- or 6-coordinate) intermediates
- Undergo rapid stereochemical inversion
Such flexibility is characteristic of organogermanium chemistry, which sits between silicon and tin in reactivity and bonding behavior.
Implication:
Stable chiral germanium compounds often require:
- Bulky substituents
- Structural constraints
4. Types of Chirality in Germanium Systems
(a) Central Chirality: Classical Ge stereocenter (GeR₁R₂R₃R₄)
(b) Axial and Helical Chirality: Occurs in systems with restricted rotation; Observed in extended frameworks or bulky substituent environments
(c) Advanced Chirality Types: Recent research shows germanium can exhibit: Ge-stereogenic centers, C-stereogenic germanes Planar, and axial chirality. Modern catalytic methods now access this full spectrum of stereochemical architectures.
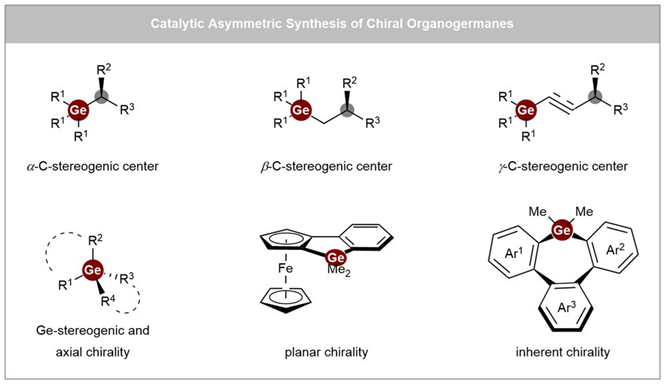
5. Synthesis of Chiral Germanium Compounds
Historically, synthesizing chiral germanium compounds has been difficult due to limited synthetic accessibility.
Traditional methods:
- Resolution of racemates
- Use of chiral auxiliaries
Modern breakthroughs (2024–2026):
Recent studies highlight a shift toward asymmetric catalysis, including:
- Transition-metal-catalyzed hydrogermylation
- Carbene insertion into Ge–H bonds
- Enantioselective coupling reactions
- Desymmetrization strategies
A notable 2025 study demonstrated a four-step synthesis from GeCl₄ to chiral germanium centers, combining deborylative alkylation with catalytic desymmetrization.
6. Applications: Why Germanium Chirality Matters
(a) Medicinal Chemistry
Organogermanium compounds are being explored as:
- Drug candidates
- Bioisosteres (carbon/silicon replacements)
They can serve both as therapeutic agents and synthetic intermediates in drug development.
(b) Materials Science
Germanium plays a key role in: Semiconductor technology, Optoelectronic materials
Chiral germanium systems may enable: Circularly polarized light devices, Chiral electronic materials
(c) Synthetic Chemistry
Organogermanes are increasingly used as: Coupling partners, Reactive intermediates. Their reactivity lies between organosilicon and organotin compounds, offering unique synthetic advantages.
7. Comparing Carbon vs Germanium Chirality
| Feature | Carbon | Germanium |
| Atomic size | Small | Larger |
| Chirality stability | Very high | Moderate |
| Inversion barrier | High | Lower |
| Hyper coordination | Rare | Common |
| Synthetic maturity | Advanced | Emerging |
Key Insight
Germanium expands the definition of chirality from a static, rigid concept (carbon) to a dynamic, flexible stereochemical system.
8. Why Don’t We Have Chiral Germanium Drugs Yet? ⭐
With such promise, a natural question arises:
If germanium can be chiral—and we can now synthesize it—why hasn’t it reached the clinic?
i. Configurational instability
- Easy inversion → racemization
- Hard to maintain enantiopurity in biological systems
ii. Toxicity concerns (historical)
- Some germanium compounds showed nephrotoxicity
- Slowed pharmaceutical adoption
iii. Synthetic maturity (until recently)
- Limited access to stable enantiopure compounds
👉 Notice how this connects back:
- Section 3 → instability
- Section 5 → synthetic limitations
🧠 Chiralpedia Insight
In drug design, chirality must not only exist—it must persist under biological conditions.
Conclusion: A New Frontier in Stereochemistry
Chiral germanium chemistry represents a compelling extension of classical stereochemistry into the realm of main-group elements. Once considered synthetically inaccessible, the field is now rapidly evolving due to advances in asymmetric catalysis and mechanistic understanding. For students and researchers, germanium offers an important conceptual takeaway:
Chirality is not limited by element—it is governed by structure, symmetry, and dynamics.
As new synthetic tools emerge, germanium may play an increasingly important role in drug design, catalysis, and advanced materials, making it a valuable addition to the stereochemist’s toolkit.
References
Wikipedia contributors. (2026, April 16). Organogermanium chemistry. In Wikipedia. https://en.wikipedia.org/wiki/Organogermanium_chemistry
Liu SW, Ke J, He C. Recent advances in catalytic asymmetric synthesis of chiral organogermanes. Chiral Chem. 2026;2:202601. https://doi.org/10.70401/cc.2026.0013
Wang, K., Liu, XY. & Dong, Z. Synthesis of chiral germanium center enabled by poly-deborylative alkylation and desymmetrization. Nat Commun 16, 5013 (2025). https://doi.org/10.1038/s41467-025-60397-x
Ai-Cui Han, Li-Jun Xiao, and Qi-Lin Zhou. Construction of Ge-Stereogenic Center by Desymmetric Carbene Insertion of Dihydrogermanes. J. Am. Chem. Soc. 2024, 146, 8, 5643–5649. https://doi.org/10.1021/jacs.3c14386
XiaoZhi Lim. Germanium gets chiral, 2024. https://cen.acs.org/synthesis/Germanium-chiral/102/web/2024/02
Further Reading