Lead
The story of chirality gained momentum in the late 19th century, as chemists began unraveling the structural principles behind molecular asymmetry. Pioneers like Jacobus Henricus van’t Hoff and Joseph Achille Le Bel laid the groundwork for modern stereochemistry by introducing three-dimensional molecular models. Their work bridged theoretical chemistry and practical applications, setting the stage for a deeper understanding of how molecular structure governs behavior in biological and chemical systems. This chapter explores the breakthroughs, challenges, and legacies of these scientific visionaries.
Jacobus Henricus van’t Hoff: Introducing the Third Dimension
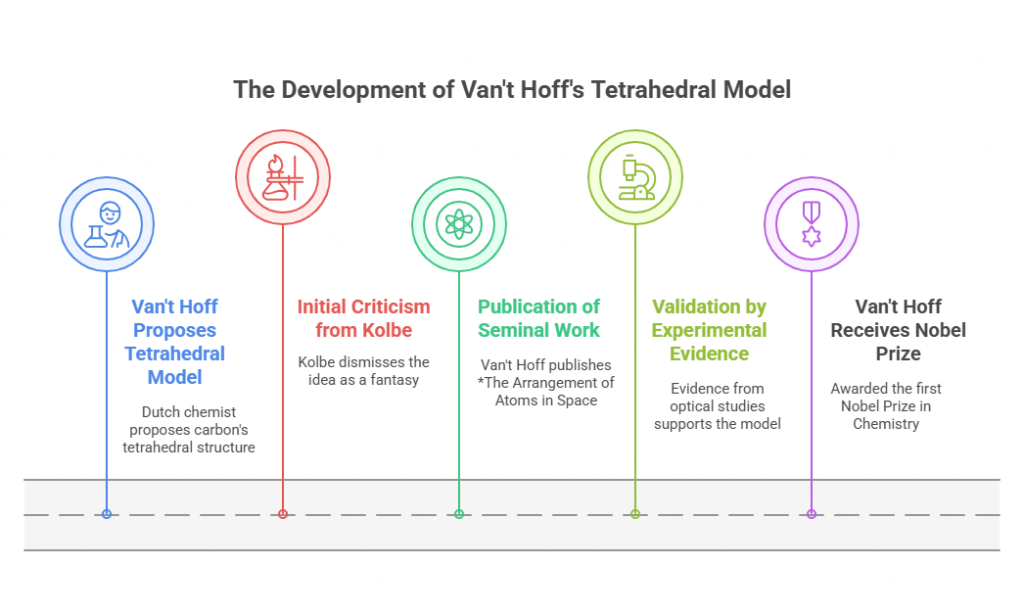
In 1874, Dutch chemist Jacobus Henricus van’t Hoff proposed a revolutionary idea: that carbon atoms are tetrahedral, bonding with four substituents in three-dimensional space. This bold hypothesis explained the existence of stereoisomers—compounds with the same chemical formula but different spatial arrangements. Van’t Hoff’s tetrahedral model elegantly accounted for the optical activity observed in chiral molecules.
Van’t Hoff’s work faced immediate criticism. Chemistry at the time was rooted in two-dimensional structural representations, and his proposal seemed speculative and abstract. Influential chemists such as Hermann Kolbe dismissed van’t Hoff’s ideas, calling them “geometrical fantasies.” Yet van’t Hoff persisted, publishing his seminal work The Arrangement of Atoms in Space to explain his theory. Over time, experimental evidence – including optical rotation studies – validated his predictions, and his model became foundational to stereochemistry.
The implications of van’t Hoff’s work were profound. By introducing three-dimensionality into molecular representations, he opened new avenues for understanding chemical reactivity, biological interactions, and material properties. His contributions earned him the first Nobel Prize in Chemistry in 1901, marking the beginning of stereochemistry as a rigorous scientific discipline.
Joseph Achille Le Bel: A Complementary Vision
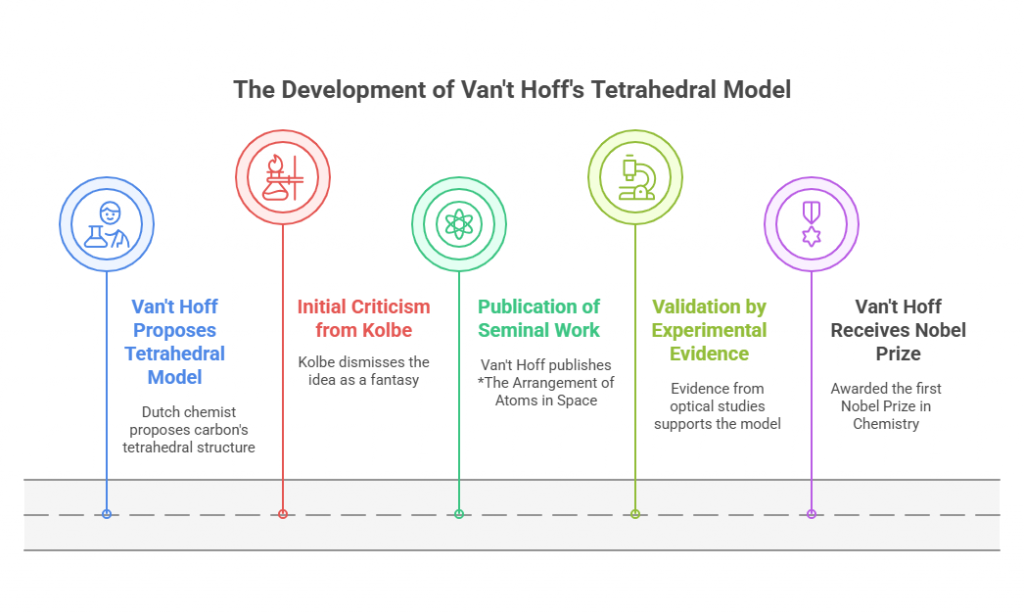
In the same year as van’t Hoff’s breakthrough, French chemist Joseph Achille Le Bel independently proposed a theory of molecular asymmetry. Le Bel focused on the relationship between molecular structure and optical activity, arguing that asymmetry arose from the spatial arrangement of substituents around chiral centers. While van’t Hoff emphasized the geometry of individual carbon atoms, Le Bel explored the broader implications of molecular asymmetry.
Le Bel’s approach was innovative but faced challenges in gaining recognition. Working in relative isolation, he struggled to disseminate his ideas widely. Nonetheless, his insights complemented van’t Hoff’s work, providing a unified framework for understanding stereoisomerism. Together, their theories established the concept of chiral centers and set the stage for future advancements in organic chemistry.
The Struggle for Acceptance
The introduction of stereochemical theories in the late 19th century was met with resistance from the scientific community. Many chemists adhered to traditional structural formulas and were skeptical of three-dimensional models. Critics argued that stereochemistry lacked empirical validation and dismissed it as speculative.
Despite these challenges, van’t Hoff and Le Bel found support among forward-thinking scientists. Advances in spectroscopy, crystallography, and polarimetry provided experimental evidence for their theories. For example, optical rotation studies confirmed the existence of stereoisomers, while X-ray crystallography later revealed the three-dimensional arrangement of atoms in chiral molecules. These technological breakthroughs gradually silenced critics and solidified stereochemistry as a cornerstone of modern chemistry.
Hermann Emil Fischer: Bridging Theory and Practice
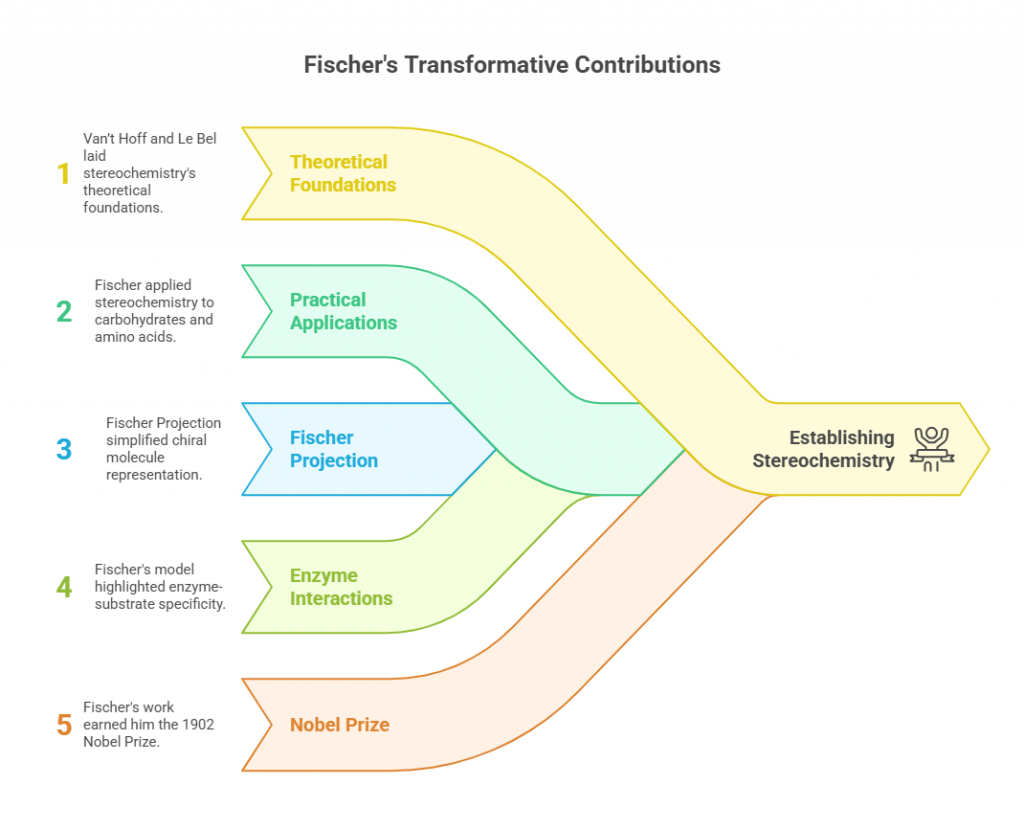
While van’t Hoff and Le Bel laid the theoretical foundations of stereochemistry, Hermann Emil Fischer brought these concepts into practical applications. Fischer’s work on carbohydrates and amino acids demonstrated how stereochemical principles could explain the behavior of complex biomolecules. In the 1890s, he introduced the Fischer projection, a simplified method for representing chiral molecules in two dimensions while preserving their stereochemical information.
Fischer’s research on sugars highlighted the importance of chirality in biological systems. He demonstrated that the stereochemistry of glucose and other carbohydrates influenced their reactivity and interaction with enzymes. Fischer’s lock-and-key model, which described the specificity of enzyme-substrate interactions, further underscored the role of molecular asymmetry in biochemistry.
Fischer’s contributions were transformative but not without challenges. Synthesizing and characterizing stereoisomers required meticulous experimentation, and the tools available to Fischer were rudimentary. Despite these difficulties, his work earned him the Nobel Prize in Chemistry in 1902 and established stereochemistry as a vital field in both organic chemistry and biochemistry.
Stereochemistry in Practice: Early Applications
By the early 20th century, stereochemistry had moved from theoretical speculation to practical application. One of the earliest successes was in the pharmaceutical industry, where the stereochemistry of drugs became a critical factor in their efficacy. For example, researchers discovered that the optical activity of certain alkaloids, such as morphine and quinine, influenced their physiological effects.
The study of stereoisomers also advanced the understanding of natural products. Chemists began isolating and characterizing chiral compounds from plants and animals, revealing the structural complexity of biomolecules. These discoveries paved the way for synthetic chemistry, where stereochemical control became essential for designing molecules with specific properties.
Challenges in Early Stereochemistry
The pioneers of stereochemistry faced numerous obstacles. Instrumentation was primitive, requiring chemists to rely on indirect methods to infer molecular structures. For example, optical rotation measurements provided evidence of chirality but did not reveal the exact arrangement of atoms. Crystallography, which would later revolutionize structural chemistry, was still in its infancy.
Moreover, the synthesis of stereoisomers posed significant challenges. Early chemists lacked the tools and techniques to selectively produce one enantiomer over another. Racemic mixtures, containing equal amounts of enantiomers, were often the default outcome of chemical reactions. Developing methods for enantioselective synthesis would become a major focus of 20th-century chemistry.
The Legacy of Early Stereochemistry
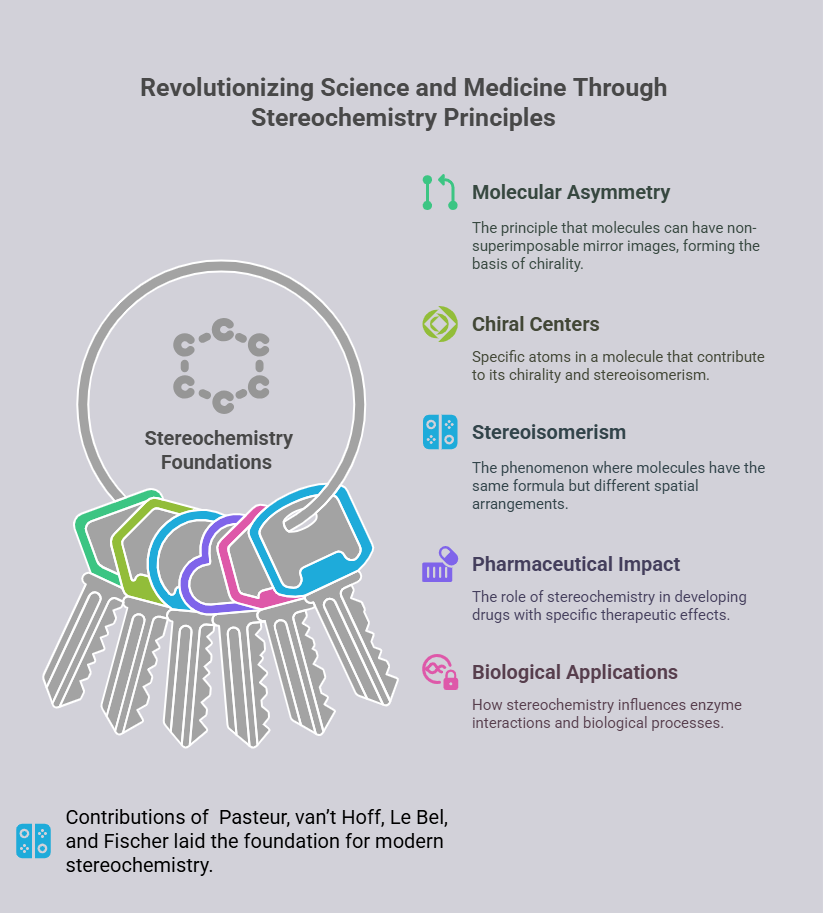
The contributions of van’t Hoff, Le Bel, and Fischer laid the foundation for modern stereochemistry, influencing a wide range of scientific disciplines. Their work established the principles of molecular asymmetry, chiral centers, and stereoisomerism, which remain fundamental to organic chemistry.
The impact of stereochemistry extends beyond academia. In the pharmaceutical industry, understanding chirality is essential for drug development. Many drugs, such as ibuprofen and albuterol (chiral drugs), exist as stereoisomers, with only one enantiomer (eutomer) providing therapeutic benefits. The ability to design and synthesize enantiomerically pure compounds has revolutionized medicine, reducing side effects and improving efficacy.
In biology, stereochemistry has illuminated the mechanisms of enzyme-substrate interactions, protein folding, and DNA replication. The lock-and-key model proposed by Fischer remains a cornerstone of enzymology, while advances in structural biology continue to reveal the stereochemical basis of life’s processes and evolved Chiral Pharmacology.
Conclusion: Building on a Solid Foundation
The building blocks of stereochemistry, introduced by van’t Hoff, Le Bel, and Fischer, represent a triumph of scientific imagination and perseverance. Their contributions bridged theoretical models and practical applications, transforming our understanding of molecular structure and its role in chemistry and biology.
As we delve deeper into the world of chirality, the legacy of these pioneers remains evident. Their work not only provided the framework for modern stereochemistry but also inspired generations of scientists to explore the complexities of molecular asymmetry. From drug development to materials science, the principles they established continue to shape the frontiers of research and innovation.
References and Further Reading
van’t Hoff JH (1874). “The Arrangement of Atoms in Space.” Archives Neerlandaises des Sciences Exactes et Naturelles.
Le Bel JA (1874). “On the Relations Which Exist Between Atomic Formulas of Organic Compounds and the Rotatory Power of Their Solutions.” Bulletin de la Société Chimique de France.
Fischer E (1890). “On the Configuration of Grape Sugar and Its Isomers.” Berichte der Deutschen Chemischen Gesellschaft.
Brown HC (1977). “The Development of Asymmetric Synthesis.” Journal of Organic Chemistry. 42(10): 1399-1407.
Ernest L. Eliel, Samuel H. Wilen, Stereochemistry of Organic Compounds, 1994, Wiley. ISBN: 978-0-471-01670-0 https://www.wiley.com/en-in/Stereochemistry+of+Organic+Compounds-p-9780471016700
Alan Bassindale, The third dimension in organic chemistry, Chichester : John Wiley and Sons , 1984. ISBN: 047190189X