L-(+)-Ascorbic Acid is another old chiral drug from natural source. It is a water-soluble vitamin (Vitamin C). Ascorbic acid is a potent reducing and antioxidant agent that functions in fighting bacterial infections, in detoxifying reactions, and in the formation of collagen in fibrous tissue, teeth, bones, connective tissue, skin, and capillaries.
Found in citrus and other fruits, and in vegetables, Vitamin C cannot be produced or stored by humans and must be obtained in the diet. Ascorbic acid was isolated by the Hungarian biochemist Albert Szent-Györgyi (1893–1986) from fruit juices in 1928 and he was awarded the Nobel Prize in physiology or Medicine in 1937 (https://www.nobelprize.org/prizes/medicine/1937/summary/), partly for this work,. In the very same year the English chemist Walter Norman Haworth (1883–1950) established the structure and also provided a synthesis for ascorbic acid. For this contribution one half of the Nobel Prize in Chemistry went to him (https://www.nobelprize.org/prizes/chemistry/1937/summary/).
Chirality and biological activity
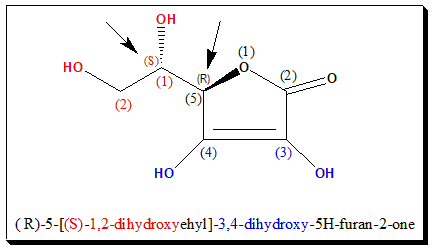
Ascorbic acid carries two stereogenic centers and exists in 4 stereoisomeric forms. Out of the possible 4 stereoisomers (2 enantiomeric pairs), the L-(+)- Ascorbic acid exhibits the redox activity and can be used to treat scurvy. The stereogenic centers are indicated by arrows.
Therapeutic category
Anti-scurvy, antioxidant, Dietary supplement
Exercise
Draw the structure of other three stereoisomers of Ascorbic acid, assign configuration and study their stereoisomeric relationship
References:
Francotte, Eric; Lindner, Wolfgang. Chirality in drug research. Eric Francotte, W. Lindner. Weinheim: Wiley-VCH. ISBN 978-3-527-60943-7. Page 3-25, 2006.
“The handed world“, https://chiralpedia.com/blog/the-handed-world/.
“Chiral twins – Identical? … But not really!”, https://chiralpedia.com/blog/ chiral-twins-identical-but-not-really/.
Harkishan Singh and V.K. Kapoor. Medicinal chemistry and pharmaceutical chemistry, Vallabh Prakashan, New Delhi, Pages 723-25, 2012.

